What Really causes eczema? Examining the science behind the itch
Eczema is more than just dry itchy skin. It's the result of a complex mix of genetics, immune activity, environmental triggers, and gut health. Here’s what the latest science tells us about how it starts and why it lingers.
This article covers the following topics:
- Genetic susceptibility to eczema
- Skin barrier function
- Environmental factors & inflammation
- Immune regulation and barrier function
- The Gut-Skin connection
Click to skip ahead to a specific section.
Eczema, or atopic dermatitis, is often explained in simple terms: “it’s inherited,” “it’s autoimmune,” or “it’s just dry, sensitive skin.” But those explanations often still leave people scratching around for answers. Over the past two decades, however, new research has transformed our understanding of eczema, revealing it to be the result of a combination of genetic susceptibility, skin barrier weakness, immune imbalance, environmental exposures, and changes in the gut microbiome. In other words, eczema is not as mysterious as it once was, but it is more complex than many standard explanations suggest. This article examines the science behind these interconnected systems and explains how eczema begins, why it persists, and how the pieces fit together.
Genetic Susceptibility to Eczema
For many, a family history of eczema, asthma, or allergies is very common. Studies suggest that around 70% of individuals with atopic dermatitis have relatives affected by at least one of these conditions. When both parents have eczema, the likelihood that their child will develop it is high, with some estimates approaching 80%. Twin studies also back up the genetic component. Identical twins who share almost all of their DNA are far more likely to both have eczema than non-identical twins, who only share about half of their DNA. If one identical twin has eczema, the chance that the other twin has it can be as high as 85%. For non-identical twins, the odds are much lower, at around 20%.
Many genes have been examined in the search for a genetic explanation for eczema, but one consistently stands out: the gene that encodes filaggrin. Filaggrin is a structural protein essential for forming the outermost layer of the skin. As skin cells mature and move toward the surface, filaggrin helps pack them tightly together, turning them into strong, flattened cells that form the stratum corneum (the outer layer of the skin). It also breaks down into smaller molecules that help the skin retain moisture and maintain a slightly acidic surface, which supports barrier strength and discourages harmful microbes.
Some people carry “loss-of-function” mutations in the filaggrin gene, meaning they produce less filaggrin. These mutations are the strongest known genetic risk factor for eczema. Inheriting one copy of a mutated filaggrin gene increases the odds of developing eczema by roughly three-to-four times compared with non-carriers. Individuals who inherit two defective copies face an even greater increase in risk, with odds rising several times higher again. Without sufficient filaggrin, the structural integrity of the outer skin layer becomes compromised, making it more prone to water loss and easier for environmental substances to enter.
Interestingly, these mutations are far more common in Northern European and other high-latitude populations. One theory suggests that slightly reduced filaggrin may have allowed more ultraviolet B (UVB) light to penetrate the skin, helping our ice-age ancestors produce enough vitamin D in regions with limited sunlight. It remains a theory, but it raises the possibility that something which once offered an advantage may now increase skin vulnerability in modern environments.
Filaggrin loss-of-function mutations do not usually eliminate filaggrin production entirely. In most people, they reduce the amount of functional filaggrin rather than stopping it altogether. This means the barrier may be weaker, but not uniformly defective across the whole body. Areas of skin that are less exposed may appear relatively normal. But regions that are frequently flexed, stretched, or exposed to the environment, such as the elbows, knees, the neck, hands, and face experience greater stress and contact with irritants. In these areas, even a modest reduction in filaggrin can be enough to tip the balance toward barrier breakdown, which explains why eczema often appears in characteristic locations rather than affecting every patch of skin equally.
Importantly though, genetics does not give us the full picture. Only around 40–50% of people with eczema carry filaggrin loss-of-function mutations, and many individuals with these mutations never develop eczema at all. Genes may set the stage, but they do not determine the outcome on their own.
Skin Barrier Integrity and Eczema
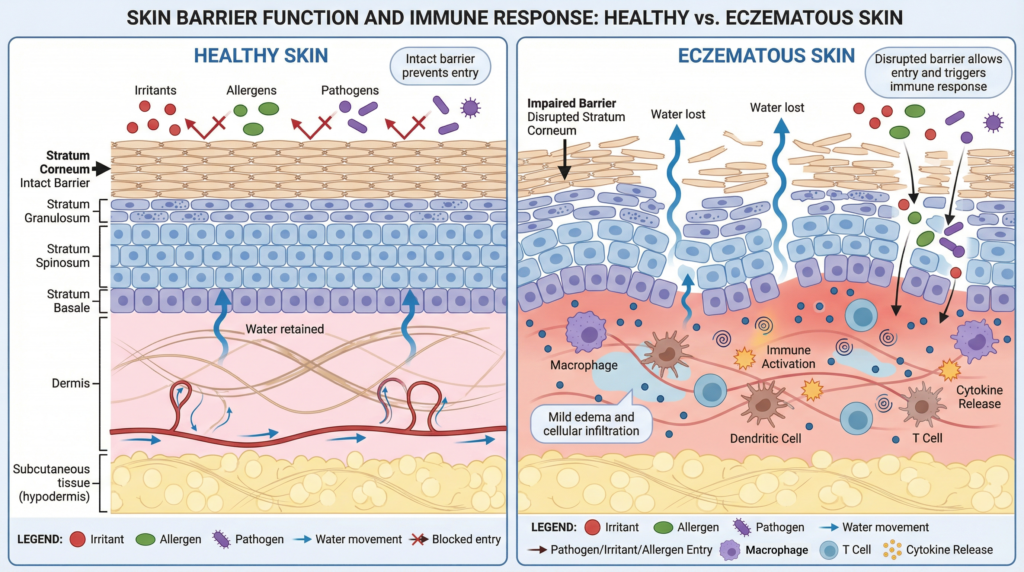
To see how genetic risk translates into visible symptoms, it helps to look more closely at the skin barrier itself. The outermost layer of the skin, known as the stratum corneum, is a tightly organised structure made up of flattened cells pressed closely together. It acts as both a physical and chemical shield, preventing too much water from escaping while blocking the entry of microbes, irritants, and allergens. Its strength depends on more than just structural proteins like filaggrin. Lipids, enzymes, and natural moisturising factors all work together to keep the barrier intact and the skin surface slightly acidic.
Filaggrin’s role becomes clearer when we examine how skin cells develop. New keratinocytes (skin cells) are formed in the deepest layer of the epidermis and gradually move upward over several weeks. As they rise, they change. In the upper layers of the skin, a large protein called profilaggrin is broken down into filaggrin, which binds to keratin fibres inside the cells and compacts them. This process turns the soft living cells into dense, flattened cells that form the outer shield of the skin. Later, filaggrin breaks down into smaller molecules that help draw water into the skin and maintain its acidity.
When filaggrin levels are reduced, whether because of genetics or other factors, this structure becomes less stable. The “mortar” between the “bricks” is weaker, and small gaps appear between cells. As a result, water escapes more easily through the surface via a process known as transepidermal water loss, which is why eczema-prone skin feels persistently dry, tight, and fragile.
Those same microscopic gaps also make it easier for external substances to enter the body. Proteins from dust mites, pollen, pet dander, harsh detergents, air pollutants, and everyday chemicals can penetrate deeper into the skin than they normally would. Once inside, they come into contact with immune cells that are not used to encountering them so directly, prompting an inflammatory response that can become difficult to resolve.
How Environmental Factors Sustain Inflammation
In healthy skin, most irritants and allergens are stopped at the surface. But when the skin barrier is weakened, they are able to penetrate into the lower layers, where they are detected by keratinocytes and resident immune cells. These cells alert the immune system, and what would otherwise be a minor exposure can become the trigger for a major inflammatory response.
Modern life has intensified this interaction. Compared to previous generations, we are now exposed to a far greater number of synthetic chemicals and processed products. Detergents in cleansers, preservatives in cosmetics, air pollutants from traffic, microplastics, and industrial compounds are now routine parts of daily life. While these substances are considered safe for the general population, they represent repeated challenges to those with impaired skin barriers that can consistently provoke and sustain inflammation.
Whilst our environment has witnessed a proliferation of novel man-made substances, our body’s own microbial landscape has changed in the opposite direction. One popular explanation for eczema, dubbed the “hygiene hypothesis”, suggests that early-life exposure to diverse microbes helps train the immune system to respond appropriately to harmless substances. In highly sanitised urban environments, children may encounter fewer of these microbial signals, and as a result, the immune system may be more likely to respond strongly to substances that would otherwise be ignored.
When you combine these two factors – increased chemical exposure and reduced microbial training, the immune system may be more easily provoked by default. In individuals with a weakened skin barrier, environmental particles gain entry more easily, and the immune response they trigger may be more exaggerated and less well regulated than in healthy individuals.
In this way, the environment does not necessarily cause eczema by itself. But when the skin barrier is weakened, everyday exposure to certain chemicals, allergens, and other substances can repeatedly trigger a heightened immune response, leading to a persistent inflammatory state.
Immune Regulation and Barrier Function
Once irritants and allergens enter the skin, the immune system responds by activating what is known as the type 2 immune pathway. This pathway is driven by molecules such as interleukin-4 (IL-4) and interleukin-13 (IL-13). These cytokines evolved to help the body defend itself against parasites, but in eczema they become activated in response to environmental irritants entering as a result of barrier disruption.
Type 2 cytokines have several effects. They promote the production of IgE antibodies, recruit inflammatory cells, and increase itch sensitivity.
It’s important to remember that itching is part of the body’s natural defence system. In healthy circumstances, an itch alerts us to potential threats like insects or irritants, and scratching helps remove them while increasing local blood flow to support immune activity and repair. But in eczema, the inflammatory signals created by IL-4 and IL-13 cytokines lower the threshold at which itch fibres fire. Nerve endings become hypersensitive, meaning even mild stimuli can provoke intense itching.
The type 2 immune pathway also reduces the production of filaggrin in skin cells, further weakening the skin barrier. Even in people without a filaggrin mutation, inflamed skin can produce less filaggrin. As the barrier weakens, more irritants enter, and as more irritants enter, the immune system responds again. Over time, this creates a vicious cycle of barrier damage and chronic inflammation creating the visible symptoms of eczema.
Persistent type 2 signalling also affects the skin’s microbiome. Protective antimicrobial peptides are reduced, making it easier for organisms such as Staphylococcus aureus to colonise the skin. This bacterium produces toxins and enzymes that further damage the barrier and stimulate additional immune activation, causing more scratching, which leads to further barrier damage.
What begins as a protective immune response gradually intensifies into a state of ongoing inflammation. But importantly, this immune activity is not confined to the skin. A large proportion of immune cells reside in the gut, where they are constantly interacting with food fragments and other microbial signals from the outside world. Inflammatory signals generated there can circulate throughout the body and influence how strongly (or how calmly) the immune system reacts elsewhere, such as in the skin.
The Gut–Skin Connection
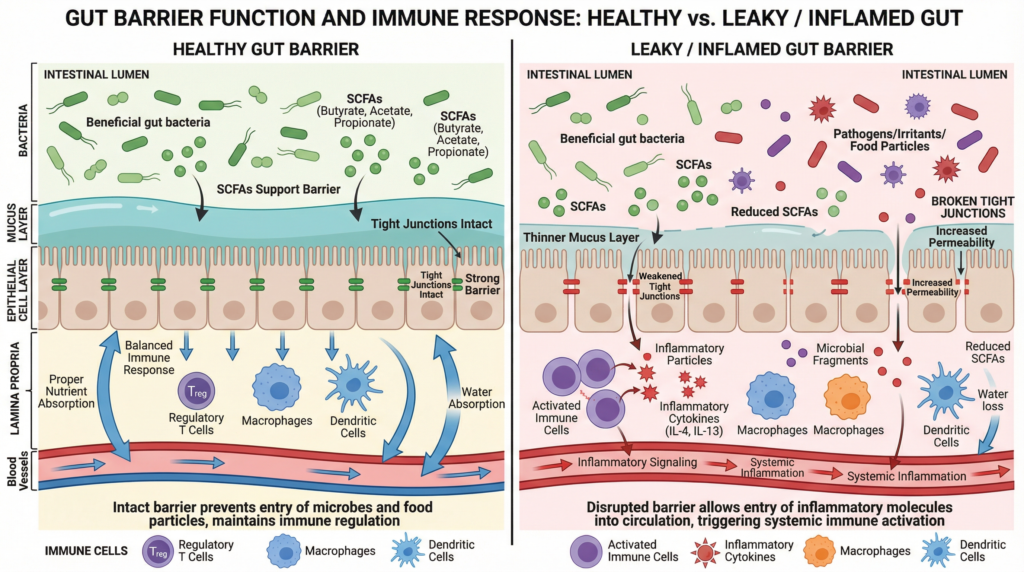
The lining of the gut performs a similar function to the skin barrier. It separates the internal environment from a constant stream of food particles, microbes, and microbial by-products. Like the skin, it is densely populated with immune cells. In fact, a significant proportion of the body’s immune system resides in the gastrointestinal tract, where it is continuously interpreting signals and deciding whether to mount an inflammatory response or remain calm.
Living within the gut is the microbiome – trillions of bacteria that coexist with us. From early life onward, these organisms help to shape immune development and calibrate how reactive the immune system becomes. Certain bacterial groups are consistently associated with healthier immune regulation. For example, species such as Bifidobacterium and Lactobacillus, are often found in higher abundance in infants who do not go on to develop eczema. In adults, reduced levels of short-chain fatty acid–producing bacteria such as Faecalibacterium prausnitzii have also been reported in individuals with atopic dermatitis.
The reason these microbes matter is because of what they produce. When gut bacteria ferment dietary fibre, they generate short-chain fatty acids (SCFAs) such as butyrate, acetate, and propionate. These compounds strengthen the gut lining, support energy production in intestinal cells, and help maintain tight junctions between cells. SCFAs also promote the production of regulatory T cells – immune cells that help prevent excessive inflammation and keep type 2 immune responses under control.
When levels of SCFA-producing bacteria are reduced, these regulatory signals may weaken. The gut lining can become more permeable, allowing microbial fragments and inflammatory molecules to enter circulation more easily. These circulating particles can then interact with immune cells elsewhere in the body (including in the skin), shifting the body into an inflammatory state.
This is where the connection to the skin becomes particularly important. As described in the section above, type 2 cytokines such as IL-4 and IL-13, whether activated in the skin itself or influenced by systemic immune signals, are known to directly reduce filaggrin production in skin cells, even in people without a filaggrin gene mutation. This means that increased intestinal permeability, otherwise known as a “leaky gut”, can trigger an immune response which has a direct impact on the skin barrier, and may play a pivotal role in causing eczema.
Data from a 2025 study backs this up. It found that cases of more severe eczema were associated with lower levels of beneficial gut-derived metabolites and higher levels of biomarkers linked to increased intestinal permeability, adding weight to the idea that gut imbalance may contribute to the overall inflammatory load in eczema.
The gut and the skin are not isolated systems. Both are barrier organs. Both are shaped by microbial communities. Both feed into the same immune network. When microbial balance in the gut shifts and regulatory signals decline, systemic inflammation may become easier to trigger on the skin.
Bringing it all together: What causes eczema?
Research increasingly shows that eczema does not begin with one single fault in the body. Instead, it develops through the interaction of several systems: genetic susceptibility, the strength of the skin barrier, the way the immune system behaves, environmental exposure, and the balance of microbes in the gut. For many people, these influences begin shaping their risk of developing eczema very early on in life.
Genetics can influence how resilient the skin barrier is from birth. People who carry filaggrin loss-of-function mutations start life with skin that may be structurally less robust. But genes alone do not decide what will happen. Many people with eczema do not carry a filaggrin mutation, and many people who do carry one never develop symptoms. This tells us that inherited risk usually needs to combine with other factors before eczema appears.
One of those factors may be the early-life gut microbiome. During the first months and years of life, the gut is still being colonised by bacteria, which are seeded in the gut from birth. In fact, the way a baby is born can heavily influence the initial composition of the gut microbiome. Babies delivered vaginally are exposed to their mother’s vaginal and gut bacteria, while those born by caesarean section tend to acquire a different mix of microbes. Antibiotic exposure during pregnancy, delivery, or shortly after birth can further disrupt this early colonisation, preventing helpful bacteria from taking hold in the gut. A large study on infants recently found that delivery method, feeding type, and early antibiotic use strongly shaped the gut microbiome during the first year of life, and certain microbial patterns were consistent with the development of eczema later in childhood.
In very young children, the microbiome is naturally less diverse and more unstable than in adults, and the immune system is still learning how to respond appropriately to harmless substances. If the microbial balance is disturbed during this period, immune regulation may be affected. Lower levels of beneficial bacteria and reduced production of short-chain fatty acids, compounds that help regulate inflammation, can weaken the signals that normally keep immune responses under control. In this setting, type 2 immune responses may become more easily amplified, increasing the likelihood that eczema will develop, especially in children who have a genetic susceptibility to eczema.
This early window may help explain why eczema is so common in childhood. In many industrialised countries, around 10–20% of children are affected. But in adulthood, the numbers are lower, often closer to 2–10% depending on the population studied. Many children see their symptoms improve or disappear over time. One possible explanation is that as the gut microbiome matures and becomes more diverse, immune regulation improves. A richer microbial community and stronger short-chain fatty acid production may help dampen excessive type 2 inflammation, allowing the skin barrier to stabilise.
Even in adulthood, however, immune balance is never fixed. Lifestyle factors continue to influence the gut and the immune system. Diets low in fibre and high in ultra-processed foods may reduce the production of beneficial microbial metabolites. Chronic stress has been shown to affect immune signalling and has been linked to changes in gut bacteria. Poor sleep and low levels of physical activity can also contribute to a more inflammatory state. In people who are susceptible, these factors may contribute to eczema persisting into adulthood or appearing for the first time, even in the absence of a filaggrin mutation.
Environmental exposures also continue to play a role. Air pollution, harsh detergents, fragranced personal care products, and occupational irritants can repeatedly stress the skin barrier. Even people without strong genetic risk may experience barrier disruption and immune activation if exposure is frequent or intense enough.
Taken together, the research suggests that eczema is not simply a surface problem. It reflects an imbalance across several bodily systems. Genetic predisposition may make the skin barrier more fragile. Changes in the gut microbiome, particularly early in life, may influence how reactive the immune system becomes, initiating type 2 inflammatory signals that can then suppress filaggrin production, weakening the barrier further. Environmental exposures can then penetrate more easily, sustaining inflammation. Each part of the process reinforces the rest.
This broader understanding does not replace standard treatments for eczema which tend to only focus on reducing inflammation on the skin. But it does suggest that long-term management may benefit from looking beyond the skin alone. Supporting gut health through diet, avoiding unnecessary antibiotics where possible, staying physically active, managing stress, and reducing exposure to harsh environmental irritants all appear to influence the inflammatory background in which eczema develops, even in individuals with filaggrin mutations.
There is reason for cautious optimism in this view. If eczema reflects an ongoing interaction between the skin barrier, the immune system, microbes, and the environment, then it is not simply a fixed genetic fate. Managing eczema may involve not only calming inflammation on the surface, but also supporting balance within the systems that shape immune responses throughout the body.
Understanding the Gut-Skin axis
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
The effect of & Nutrition on Inflammation
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
References
- Brown, S.J. and McLean, W.H.I. (2012) ‘One remarkable molecule: filaggrin’, Journal of Investigative Dermatology, 132(3 Pt 2), pp. 751–762.
- Irvine, A.D., McLean, W.H.I. and Leung, D.Y.M. (2011) ‘Filaggrin mutations associated with skin and allergic diseases’, New England Journal of Medicine, 365(14), pp. 1315–1327.
- Palmer, C.N.A., Irvine, A.D., Terron-Kwiatkowski, A., Zhao, Y., Liao, H., Lee, S.P., Goudie, D.R., Sandilands, A., Campbell, L.E., Smith, F.J.D., O’Regan, G.M., Watson, R.M., Cecil, J.E., Bale, S.J., Compton, J.G., DiGiovanna, J.J., Fleckman, P., Lewis-Jones, S., Arseculeratne, G., Sergeant, A., Munro, C.S., El Houate, B., McElreavey, K., Halkjaer, L.B., Bisgaard, H., Mukhopadhyay, S., McLean, W.H.I. and Brown, S.J. (2006) ‘Common loss-of-function variants of the epidermal barrier protein filaggrin are a major predisposing factor for atopic dermatitis’, Nature Genetics, 38(4), pp. 441–446.
- Thyssen, J.P., Godoy-Gijon, E., Elias, P.M. and Ghadially, R. (2014) ‘Evidence that loss-of-function filaggrin gene mutations evolved in Northern Europeans to favour intracutaneous vitamin D3 production’, Evolutionary Applications, 7(3), pp. 366–375.
- Thyssen, J.P., Kezic, S., Saunders, S.P., Irvine, A.D., McLean, W.H.I., Flohr, C., De Bruin-Weller, M.S., Bulfone-Paus, S., von Hertzen, L., Weidinger, S., Olesen, A.B., Carlsen, B.C., Andersen, K.E., Svensson, A., Vestergaard, C., Wallmeyer, L., Ståhle, M., Langan, S.M., Brown, S.J., Simpson, E.L. and Silverberg, J.I. (2010) ‘Loss-of-function mutations in the filaggrin gene and risk of atopic dermatitis: a meta-analysis’, British Journal of Dermatology, 163(1), pp. 1–12.
- Kumar, V., et al. (2021) ‘Filaggrin gene mutations with special reference to atopic dermatitis’, Clinical, Cosmetic and Investigational Dermatology, 14, pp. 1455–1465. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC7880084/
- Bisgaard, H., Simpson, A., Palmer, C.N.A., Bonnelykke, K., McLean, I., Mukhopadhyay, S., Pipper, C.B., Halkjaer, L.B., Lipworth, B., Hacon, S. and Custovic, A. (2008) ‘Gene–environment interaction in the onset of eczema in infancy: filaggrin loss-of-function mutations enhanced by neonatal cat exposure’, PLoS Medicine, 5(6), e131. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC2504043/
- Elias, P.M. and Steinhoff, M. (2008) ‘“Outside-to-inside” and “inside-to-outside” pathogenic mechanisms in atopic dermatitis’, Journal of Investigative Dermatology, 128(5), pp. 1067–1070.
- Howell, M.D., Kim, B.E., Gao, P., Grant, A.V., Boguniewicz, M., DeBenedetto, A., Schneider, L., Beck, L.A. and Leung, D.Y.M. (2007) ‘Cytokine modulation of atopic dermatitis filaggrin skin expression’, Journal of Allergy and Clinical Immunology, 120(1), pp. 150–155. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC2669594/
- McAleer, M.A. and Irvine, A.D. (2013) ‘The multifunctional role of filaggrin in allergic skin disease’, Journal of Allergy and Clinical Immunology, 131(2), pp. 280–291.
- Kuo, I.H., Yoshida, T., De Benedetto, A. and Beck, L.A. (2021) ‘Keratinocytes: innate immune cells in atopic dermatitis’, Clinical and Experimental Immunology, 204(3), pp. 296–305.
- Lambrecht, B.N. and Hammad, H. (2019) ‘Epithelial barrier repair and prevention of allergy’, Clinical and Experimental Allergy, 49(6), pp. 743–754. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC6436854/
- Gandhi, N.A., Bennett, B.L., Graham, N.M.H., Pirozzi, G., Stahl, N. and Yancopoulos, G.D. (2016) ‘Targeting key proximal drivers of type 2 inflammation in disease’, Nature Reviews Drug Discovery, 15(1), pp. 35–50.
- Leung, D.Y.M., Boguniewicz, M., Howell, M.D., Nomura, I. and Hamid, Q.A. (2004) ‘New insights into atopic dermatitis’, Nature Reviews Immunology, 4(11), pp. 902–912.
- Nakatsuji, T., Chen, T.H., Butcher, A.M., Trzoss, L.L., Nam, S.J., Shirakawa, K.T., Zhou, W., Oh, J., Otto, M., Fenical, W. and Gallo, R.L. (2016) ‘A commensal strain of Staphylococcus epidermidis protects against skin neoplasia’, Journal of Clinical Investigation, 126(6), pp. 2335–2343.
- Rook, G.A.W. (2010) ‘Regulation of the immune system by biodiversity from the natural environment: the “Old Friends” hypothesis’, Clinical and Experimental Immunology, 160(1), pp. 70–79.
- Strachan, D.P. (1989) ‘Hay fever, hygiene, and household size’, BMJ, 299(6710), pp. 1259–1260.
- Zheng, H., Liang, H., Wang, Y., Miao, M., Shi, T., Yang, F., Liu, E. and Yuan, W. (2023) ‘Development of the early-life gut microbiome and associations with eczema in a prospective cohort’, mSystems, 8(3). Available at: https://journals.asm.org/doi/10.1128/msystems.00521-23
- Wang, Q., et al. (2019) ‘Association between the intestinal microbiota and allergic sensitization, eczema, and asthma: a systematic review’, Journal of Allergy and Clinical Immunology, 143(2), pp. 467–485.
- Lee, S.Y., et al. (2024) ‘Gut dysbiosis and adult atopic dermatitis: a systematic review’, Nutrients, 16(2). Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC11721037/
- Arpaia, N., Campbell, C., Fan, X., Dikiy, S., van der Veeken, J., deRoos, P., Liu, H., Cross, J.R., Pfeffer, K., Coffer, P.J. and Rudensky, A.Y. (2013) ‘Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation’, Nature, 504(7480), pp. 451–455.
- Salem, I., Ramser, A., Isham, N. and Ghannoum, M.A. (2018) ‘The gut microbiome as a major regulator of the gut–skin axis’, Frontiers in Microbiology, 9, 1459.
- Kim, J., Kim, B.E. and Leung, D.Y.M. (2024) ‘Skin and gut microbiome in atopic dermatitis: mechanisms and therapeutic opportunities’, Frontiers in Microbiology, 15.
- Zhang, L., et al. (2024) ‘The gut–skin axis: a bi-directional microbiota-driven relationship with therapeutic potential’, Nutrients, 16(1).
- Kim, H.J., et al. (2025) ‘Severity of atopic dermatitis is associated with gut-derived metabolites and leaky gut-related biomarkers’, Scientific Reports, 15, Article 9520.
- Nutten, S. (2015) ‘Atopic dermatitis: global epidemiology and risk factors’, Annals of Nutrition and Metabolism, 66(Suppl 1), pp. 8–16.
- Silverberg, J.I. and Hanifin, J.M. (2013) ‘Adult eczema prevalence and associations’, Journal of Investigative Dermatology, 133(11), pp. 2477–2484.
Disclaimer
The information provided on this website is for educational purposes only and is not intended as medical advice. Always consult a qualified healthcare professional before making changes to your diet, supplements, or treatment plan.
Important Links
Contact
About
Privacy Policy
Terms & Conditions